Magnetic microbead occlusion mouse glaucoma model All experiments were performed under general anesthesia using 2% isoflurane (0.8 l/min), except for electroretinogram (ERG) recordings (see below). Surgical procedures were carried out in female B6.Cg.Tg 2Jrs/J mice (Jackson Laboratory, Bar Harbor, ME) or wild-type littermate controls (3 ̶ 4 months of age) maintained under 12 h light/12 h dark cyclic light conditions with an average in-cage illumination level of 10 lx. These findings highlight the importance of restoring energy homeostasis to recover neuronal function in glaucoma.Īll animal procedures were approved by the Centre de Recherche du Centre Hospitalier de l’Université de Montréal (CRCHUM) Animal Care Committee and the Canadian Council on Animal Care guidelines. Our data reveal that AMPK is a critical mediator of RGC dendrite pathology and synapse elimination during ocular hypertension-induced metabolic stress. 
However, the role of AMPK in RGC dendritic structure and connectivity is currently unknown. Recent work has strongly implicated energy deficits in glaucoma, and reduced AMPK correlates with anti-inflammatory responses and RGC protection. Retraction of RGC dendrites with synapse disassembly is a key feature of ocular hypertension damage. The selective death of RGC is a crucial element in the pathophysiology of glaucoma, the leading cause of irreversible blindness worldwide. To address this knowledge gap, we focused on retinal ganglion cells (RGC), a population of long-projecting neurons that link the retina to the brain. 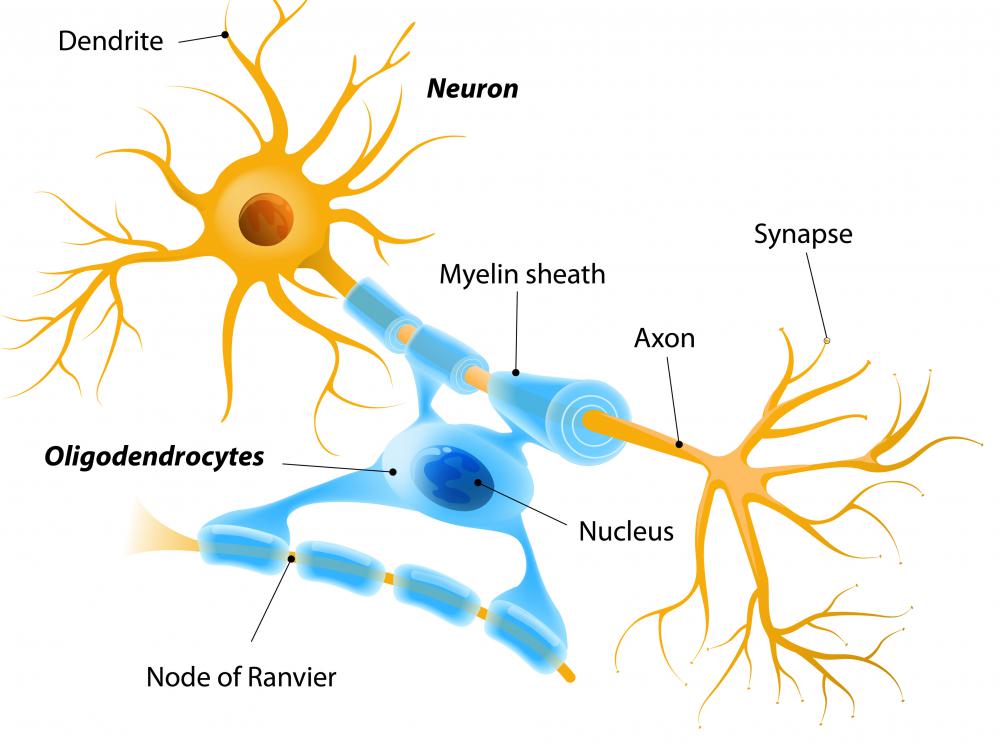
Furthermore, the role of AMPK in dendritic and synaptic alterations in injured neurons is not well understood. Although sensing ATP is critical to restore energy, the consequence of persistent AMPK activity in neurons with compromised energy supply is unknown. For example, AMPK activation during developmental energetic stress inhibits neuronal polarization and axonal outgrowth while it regulates synaptic remodeling in aging neurons. Active AMPK restores energy homeostasis through catabolic pathways that produce ATP while inhibiting processes that consume energy. During energy stress, intracellular ATP levels fall and AMP rises leading to AMPK activation. 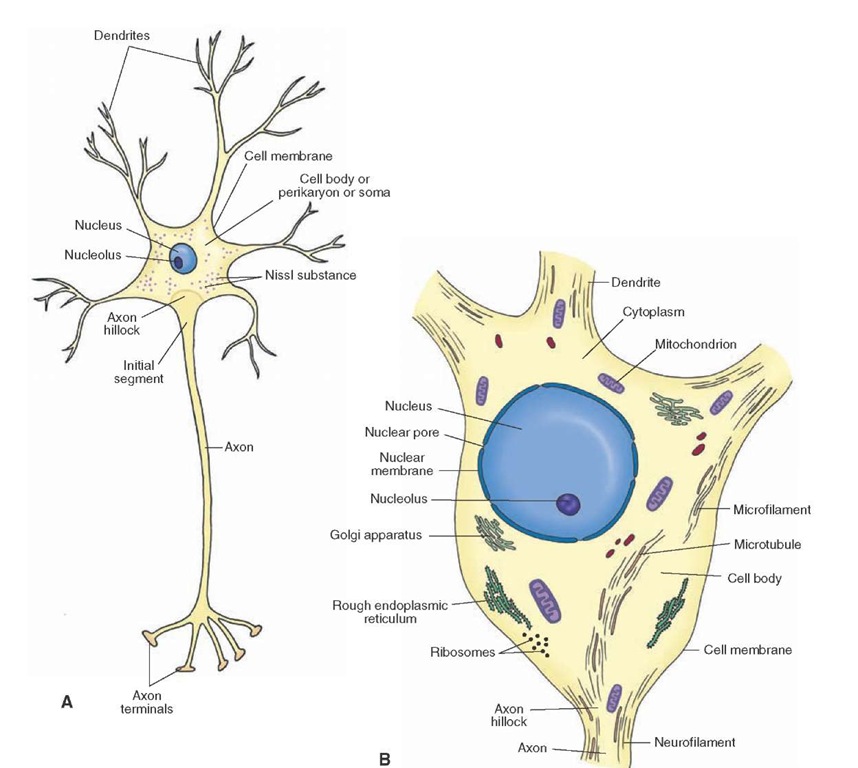
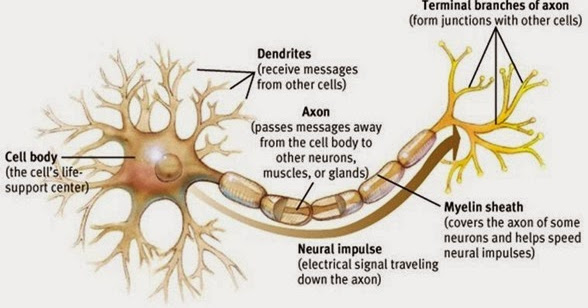
AMPK is a heterotrimeric serine/threonine kinase composed of a catalytic α subunit (PRKAA1), which is essential for AMPK function through phosphorylation of its activation loop, and regulatory β and γ subunits. However, the molecular mechanisms linking energy shortage with dendrite and synapse instability in neuropathological conditions are poorly understood.Īdenosine monophosphate-activated protein kinase (AMPK) is a highly conserved energy sensor and an important metabolic regulator. A better understanding of how neurons sense and respond to metabolic stress will provide valuable insights into strategies aimed to prevent synaptic deficits and restore neuronal circuit function. Mitochondrial dysfunction has been reported in a number of neurodegenerative diseases and correlates with dendritic pathology and synapse disassembly. When mitochondria are compromised, they produce less ATP leading to energetic stress, which is likely to affect dendrites and their synapses. The predicted energy expenditure at dendrites is substantial, consistent with the high density of mitochondria and oxidative activity found in the dendritic compartment. This study identifies AMPK as a critical nexus between bioenergetic decline and RGC dysfunction during pressure-induced stress, and highlights the importance of targeting energy homeostasis in glaucoma and other neurodegenerative diseases.Ī substantial amount of the energy produced in the brain is used for synaptic transmission. Strikingly, AMPK depletion promotes recovery of light-evoked retinal responses, improves axonal transport, and extends RGC survival. Attenuation of AMPK activity restores mTORC1 function and rescues dendrites and synaptic contacts. We show that the harmful effect of AMPK is exerted through inhibition of the mammalian target of rapamycin complex 1 (mTORC1). Our data demonstrate that AMPK triggers RGC dendrite retraction and synapse elimination. We show that adenosine monophosphate-activated protein kinase (AMPK), a conserved energy biosensor, is strongly activated in RGC from mice with ocular hypertension and patients with primary open angle glaucoma. Recent work has implicated energy deficits in glaucoma, and retinal ganglion cell (RGC) dendritic pathology and synapse disassembly are key features of ocular hypertension damage. Bioenergetic decline is a prominent feature of chronic neurodegenerative diseases, yet the signaling mechanisms that link energy stress with neuronal dysfunction are poorly understood. The maintenance of complex dendritic arbors and synaptic transmission are processes that require a substantial amount of energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
